10-Oct-2009
Direct Reduced Iron (DRI) is obtained by reducing lumps as well as fines of iron ore in solid state at a relatively low temperature of around 1000OC. A large number of DR processes are available today. SL/RN and HYL are two such DR processes. While HYL is a batch-type gas based process and uses a countercurrent shaft-furnace, the SL/RN process utilizes rotary kiln to reduce lump ore, pellets and sand iron with coal. Here we will discuss about some key features, including advantages and disadvantages of these two DR processes.
HYL III
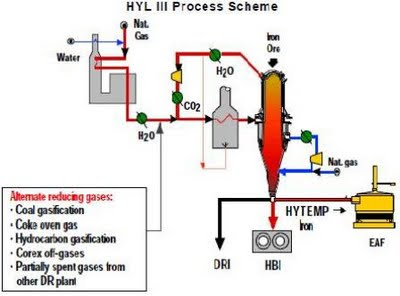
Fig: HYL III Process Scheme
The HYL process was developed in Mexico and was the forerunner of the HYL III direct reduction technology. In HYL I process, a mixture of gases containing about 89% of reducing compounds is used. Each reduction module in HYL plant consists of four units - three “in line” and the fourth in “turn around” mode. The principal change made over HYL I in HYL III was the modification of the four fixed bed reactors by a single moving bed reactor, utilizing the same gas reforming plant, auxiliary equipment and quenching towers. Actually HYL III technology is characterized by its wide flexibility for adapting to special needs, depending on available reducing gases, energy use and melt-shop requirements. Use of spent gases from direct ironmaking processes, coal gasification, energy optimization in DR plants and technology developments aimed to improve EAF productivity have been the objective of HYL. Some distinctive features of HYL III process are:
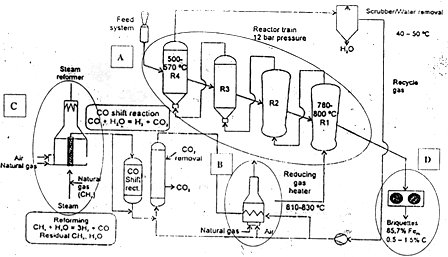
Fig: HYL III - COREX Off-Gas Process Scheme
=> The H2/CO of the reformed gas is 3, the temperature is about 930OC, the inside pressure of the countercurrent shaft-furnace is 450 kilopascals and the energy required for the reduction is basically the same as in the MIDREX process.
=> The selective elimination of H2O and CO2 from the reducing gas circuit allows maximum recycle of the reducing gases to the reduction reactor. Hence, the reducing gas make-up and the process natural gas consumption are minimized.
=> The reducing gas generation and the reduction sections of a HYL III unit are independent from an operational point of view. This feature offers important flexibility for adapting to different reducing gas sources. The process schemes based on use of alternative reducing gases from different sources and other DR/ Ironmaking sources have been proven in HYL III plants. Such alternate sources of reducing gas can be -
- Coal gasification processes.
- Coke oven gas.
- Gases from Hydrocarbon gasification.
- Partially spent gases from another DR plant.
- COREX off-gases.
=> High pressure operation (4 atmospheres or more) enables the effective control of process conditions, with smaller equipment size for gas handling and lower energy requirements (9.0 - 10.0 GJ/t).
=> The process is much flexible as far as raw material use is concerned - while it operates best with 100% pellets, even 100% lump ore of a suitable type has been used, but it is suggested to use a mixture of pellets and lump ores.
=> This technology offers the unique flexibility to produce three different product forms depending on the specific requirements of each user - Cold DRI, HBI and HYTEMP iron. Metallization can be controlled up to 95% and Carbon content 5.0%.
=> When combined with COREX off-gas as a source of reducing gas, the HYL III DR plant offers high productivity using available spent gas and benefits in steel production using HYTEMP® iron together with hot metal in EOF/BOF based steel mills.
=> The HYL III process features the flexibility of generating electric power, taking advantage of high pressure steam produced in the natural gas-steam reforming unit which can be used in a turbo generator or in a set of turbines, at a high generation capacity.
According a data of recent past, around 11 million tones of direct reduced iron (DRI) was produced in 2003 by this process in India, Grasim’s HYL plant at Raigad (Orissa) produced 0.75 million tones of HBI.
SL/RN
SL/RN is the most widely accepted coal based DR process. It was jointly developed by Stelco, Lurgi Chemie, Republic Steel Company and National Lead Corporation in 1964. In this process, the materials charged into the kiln gravitate towards the discharge end during which they are progressively heated to the temperature of reduction of around 1000 - 1100OC. The product discharged from the kiln is cooled in an extremely cooled rotary cooler around 100OC before being subjected to magnetic separation to separate sponge iron from coal ash and char. Waste gases leaving the kiln at the inlet end pass through a dust chamber and a post combustion chamber, before being cooled and cleaned in electrostatic precipitators, scrubbers or bag filters. In SL/RN technology the clean gases can be used in waste heat boilers to recover the sensible heat and the steam generated can be utilized for heating purpose or for electric power generation. Some distinctive features of SL/RN process include:
=> Flexibility with regard to the type of iron bearing materials which can be used such as lump ore, pellets, ilmanite, iron sands and steel plant wastes.
=> Use of a wide variety of solid fuels ranging from anthracite to lignite and charcoal.
=> Improved heating of the charge by submerged air injection in pre-heating zone of the kiln. This process suffers, however, from relatively big heat loss and facility size.
=> SL/RN technology provides optimized coal injection facilities at the discharge end of the kiln.
=> Waste gas conditioning by controlled post combustion followed by power generation (the power generated is more than the requirement of the plant).
The original SL/RN process has been modified in a variety of ways, particularly in India where rotary kiln DR technology has been widely applied.